Mounjaro in Leeds
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
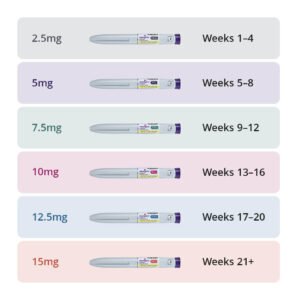
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Overview Mounjaro® (tirzepatide) is an MHRA-approved, once-weekly injection for weight management and type 2 diabetes in the UK. Available via private online pharmacies or NHS (for specific criteria), it requires…
Mounjaro Uk Mounjaro UK is a prescription medication with the active ingredient tirzepatide, used to treat adults with type 2 diabetes and for chronic weight management. It is administered as a once-weekly subcutaneous…