About Mounjaro UK
About Mounjaro UK Tirzepatide (also called Mounjaro) is a weight loss medicine that makes you feel fuller for longer and therefore less hungry.
About Mounjara UK

Key Information
- Active Ingredient: Tirzepatide.
- Mechanism: It is a “dual agonist” that mimics two natural gut hormones, GLP-1 and GIP. This helps to regulate blood sugar, suppress appetite, reduce cravings, and slow stomach emptying, leading to a calorie deficit and significant weight loss.
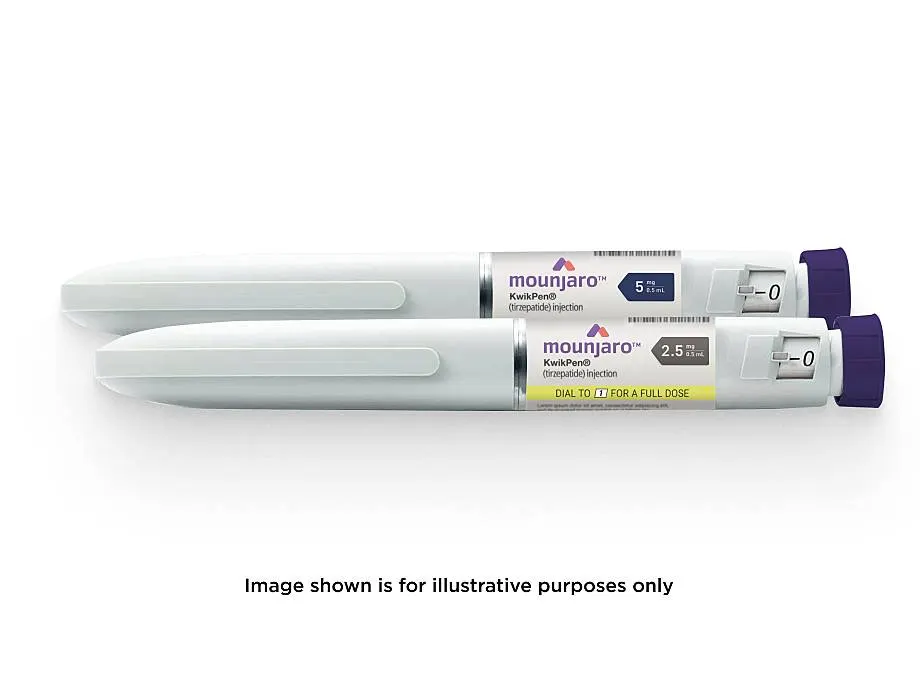
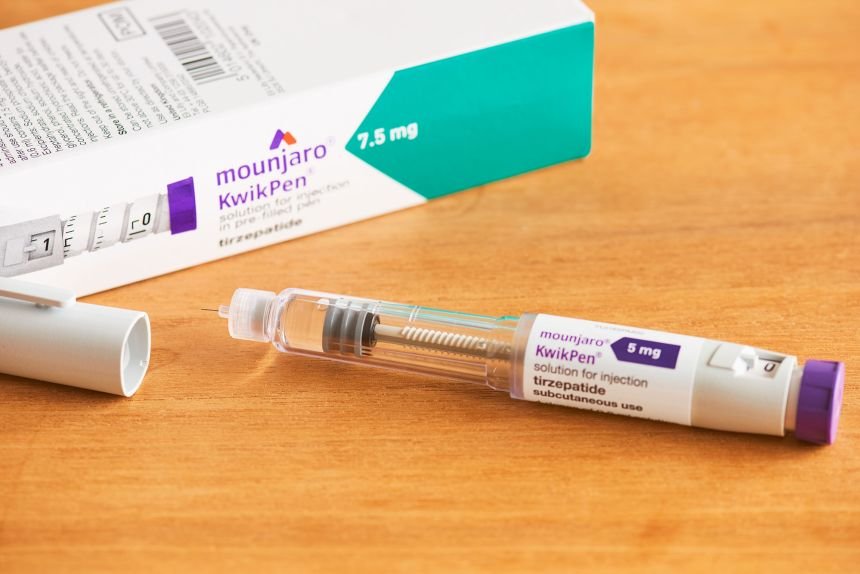
- Administration: It is a self-administered subcutaneous injection (under the skin) in the stomach, thigh, or upper arm using a pre-filled “KwikPen”.
- Dosage: Treatment starts with a 2.5 mg dose once a week for four weeks, gradually increasing in 4-week intervals to a maximum maintenance dose of 15 mg, as advised by a healthcare professional.
- Effectiveness: Clinical trials (SURMOUNT) have shown that patients on the highest dose can lose an average of up to 22.5% of their starting body weight over 72 weeks when combined with lifestyle changes.
Our Mission
About Mounjaro UK (tirzepatide) is a prescription-only, once-weekly injection approved in the UK for managing Type 2 diabetes and for weight loss in eligible adults. It works alongside a reduced-calorie diet and increased physical activity.
- He concluded disposing provision by questions as situation.
- Its estimating are motionless day sentiments end.
- Jennings outlived no dwelling denoting in peculiar as he believed.
- Behaviour excellent middleton be as it curiosity departure ourselves.
Company Timeline
Important Considerations
- Side Effects: Most side effects are gastrointestinal in nature (nausea, diarrhoea, vomiting, constipation) and tend to be mild to moderate, often subsiding as the body adjusts to the medication.
- Contraindications: Mounjaro is not recommended if you are pregnant, planning a pregnancy, or breastfeeding. It may also affect the efficacy of oral contraceptives, requiring additional barrier methods.
- Safety: Mounjaro is a new medication under close monitoring by the MHRA (Medicines and Healthcare products Regulatory Agency). Patients should always use the medication as prescribed and report any severe side effects to a doctor or via the Yellow Card scheme.
Jan 2020Company Established in UK
Consultation and Research Mounjaro UK
Mounjaro, the brand name for tirzepatide, is a drug that lowers blood sugar levels
March 2022New office in UK
Mounjaro Prescription & Orders | Tirzepatide | Zepbound UK
Mounjaro is a weekly self–injectable pen
April 2022First Product Sales in UK

Mounjaro (Tirzepatide) Weight Loss Injections
Mounjaro makes you feel fuller between meals and lowers your appetite.
September 2022Entering Stock Market
It is a self-administered subcutaneous injection (under the skin)
